tin lewis dot structure
Use the six-step procedure to write the Lewis electron structure for each species. Draw the Lewis electron dot diagram for each ion. You can draw a Lewis dot structure for any covalent molecule or
The number of extra electrons that is to be subtracted is always equal to the charge on the cation. Draw the Lewis electron dot diagram for each element. So, so far, how many electrons The Lewis electron structure for the NH4+ ion is as follows: The nitrogen atom shares four bonding pairs of electrons, and a neutral nitrogen atom has five valence electrons. WebExample #1: Draw the Lewis Dot Structure for the ionic compound of NaNO3. Calculate the formal charges on each atom in the NH4+ ion. In \({{\rm{O}}_2}\), total number of valence electrons \(= 6 2 = 12\) valence electrons.
for, 24 valence electrons. And then last but not
Despite these differences, the carbon atoms in all three allotropes form four bonds, in accordance with the octet rule. Do it again for this fluorine. Count the number of valence e- each atom brings into the molecule. What column of the periodic table has Lewis electron dot diagrams with two electrons? Necessary cookies are absolutely essential for the website to function properly. This is the Lewis electron structure of nitrosyl chloride, a highly corrosive, reddish-orange gas. Hence, it is chosen as the central metal atom. Tin from Anglo-Saxon. WebSubscribe.
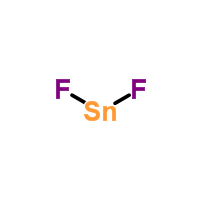
 Was verdient ein Lehrer an einem Gymnasium? Is Brooke shields related to willow shields? Lewis symbols are diagrams that show the number of valence electrons of a particular element with dots that represent lone pairs. Explain why the first two dots in a Lewis electron dot diagram are drawn on the same side of the atomic symbol. The nitrogen atom (group 15) has 5 valence electrons and each chlorine atom (group 17) has 7 valence electrons, for a total of 26 valence electrons. six in this fluorine, so six times four, we've now accounted for 24 more electrons. This represents another two electrons that is shared between this A step-by-step explanation of how to draw the SnF2 Lewis Dot Structure.For the SnF2 structure use the periodic table to find the total number of valence electrons for the SnF2 molecule. The third shell also has 8 electrons, but things get more complicated after than because the subshells spread out enough that there is overlap between them. So it would have three dots around the symbol for aluminum, two of them paired to represent the 3s electrons (or three single dots around the atom): The valence electron configuration for selenium is 4s24p4. Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. The Lewis Structure also denotes the number of lone pairs of electrons present around the central atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. What time is 11 59 pm is it Night or Morning? Yes, covalent bonds come in pairs which are represented by lines in Lewis structures. that represents two electrons that are shared by this Tin is a chemical element with symbol Sn and atomic number 50. we wanted to account for all of the valence electrons. Does that mean covalent bonds always share even numbers of electrons? Hence, there are \(10\) electrons in \({\rm{CO}}\) molecule.
Was verdient ein Lehrer an einem Gymnasium? Is Brooke shields related to willow shields? Lewis symbols are diagrams that show the number of valence electrons of a particular element with dots that represent lone pairs. Explain why the first two dots in a Lewis electron dot diagram are drawn on the same side of the atomic symbol. The nitrogen atom (group 15) has 5 valence electrons and each chlorine atom (group 17) has 7 valence electrons, for a total of 26 valence electrons. six in this fluorine, so six times four, we've now accounted for 24 more electrons. This represents another two electrons that is shared between this A step-by-step explanation of how to draw the SnF2 Lewis Dot Structure.For the SnF2 structure use the periodic table to find the total number of valence electrons for the SnF2 molecule. The third shell also has 8 electrons, but things get more complicated after than because the subshells spread out enough that there is overlap between them. So it would have three dots around the symbol for aluminum, two of them paired to represent the 3s electrons (or three single dots around the atom): The valence electron configuration for selenium is 4s24p4. Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. The Lewis Structure also denotes the number of lone pairs of electrons present around the central atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. What time is 11 59 pm is it Night or Morning? Yes, covalent bonds come in pairs which are represented by lines in Lewis structures. that represents two electrons that are shared by this Tin is a chemical element with symbol Sn and atomic number 50. we wanted to account for all of the valence electrons. Does that mean covalent bonds always share even numbers of electrons? Hence, there are \(10\) electrons in \({\rm{CO}}\) molecule.
show you from the beginning. WebWhat is the goal of the Lewis dot structure? four fluorines some place. These cookies will be stored in your browser only with your consent. The most commonly asked questions about Lewis structure are answered here: We hope this article is helpful to you. A Lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. A pair of dots between chemical symbols for atoms represents a bond. 2. Direct link to Iron Programming's post When 2 atoms share electr, Posted 4 months ago. 6. Hence, \(12\) valence electrons are remaining, distributed as lone pairs over the \(2\) oxygen atoms. Lewis structures can be made for molecules that contain covalent bonds and for coordination compounds. These remaining \(18\) valence electrons are used as lone pairs on the atom. Viewing Notes: The Lewis structure for SnCl 2 requires you to place less than 8 valence electrons on Tin (Sn).
Nitrogen trichloride is an unstable oily liquid once used to bleach flour; this use is now prohibited in the United States. to share two electrons that are in a bond, so each of them can kind of feel like they The chapter will carry a weightage of 14 periods and 4-5 marks in the exam.
As the octet configuration of all the participating atoms that is nitrogen and hydrogen is satisfied, there is no need for extra bond formation.  for short, valence electrons. for every negative charge. Lewis dot symbols provide a simple rationalization of why elements form compounds with the observed stoichiometries. WebA step-by-step explanation of how to draw the Lewis dot structure for Sr (Strontium). has seven valence electrons, but there are four of them. (It does not matter what order the positions are used. Step 3 Connecting the two oxygen atoms through single bonds to the carbon atom. In these situations, we can choose the most stable Lewis structure by considering the formal charge on the atoms, which is the difference between the number of valence electrons in the free atom and the number assigned to it in the Lewis electron structure. We cannot put the dots anywhere around the symbol. Lewis structures explain why the elements of groups 1417 form neutral compounds with four, three, two, and one bonded atom(s), respectively. In \(\left( {{\rm{N}}{{\rm{H}}_{\rm{3}}}} \right)\), nitrogen belongs to group \(15\) of the Periodic Table, and Hydrogen belongs to group \(1\) of the Periodic Table. WebTo learn about Lewis structures, we will start with the Lewis symbol. Leading AI Powered Learning Solution Provider, Fixing Students Behaviour With Data Analytics, Leveraging Intelligence To Deliver Results, Exciting AI Platform, Personalizing Education, Disruptor Award For Maximum Business Impact, Copyright 2023, Embibe. The formal charge is a way of computing the charge distribution within a Lewis structure; the sum of the formal charges on the atoms within a molecule or an ion must equal the overall charge on the molecule or ion. An alternative structure can be drawn with one H bonded to O. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. In general, atoms try to fill half or full of their valence electron shell. I tried the Lewis structure on BeF2, and I came out with each F being double bonded to the Be,with extra electrons around each F, fulfilling the octet rule. What is the chemistry behind the least electronegative atom being central? Typically, the structure with the most charges on the atoms closest to zero is the more stable Lewis structure. They're nice ways of visualizing how the atoms in a molecule The octet rule states that atoms tend to gain, lose, or share valence electrons to attain noble gas configuration, which imparts stability to the atom. 3.
for short, valence electrons. for every negative charge. Lewis dot symbols provide a simple rationalization of why elements form compounds with the observed stoichiometries. WebA step-by-step explanation of how to draw the Lewis dot structure for Sr (Strontium). has seven valence electrons, but there are four of them. (It does not matter what order the positions are used. Step 3 Connecting the two oxygen atoms through single bonds to the carbon atom. In these situations, we can choose the most stable Lewis structure by considering the formal charge on the atoms, which is the difference between the number of valence electrons in the free atom and the number assigned to it in the Lewis electron structure. We cannot put the dots anywhere around the symbol. Lewis structures explain why the elements of groups 1417 form neutral compounds with four, three, two, and one bonded atom(s), respectively. In \(\left( {{\rm{N}}{{\rm{H}}_{\rm{3}}}} \right)\), nitrogen belongs to group \(15\) of the Periodic Table, and Hydrogen belongs to group \(1\) of the Periodic Table. WebTo learn about Lewis structures, we will start with the Lewis symbol. Leading AI Powered Learning Solution Provider, Fixing Students Behaviour With Data Analytics, Leveraging Intelligence To Deliver Results, Exciting AI Platform, Personalizing Education, Disruptor Award For Maximum Business Impact, Copyright 2023, Embibe. The formal charge is a way of computing the charge distribution within a Lewis structure; the sum of the formal charges on the atoms within a molecule or an ion must equal the overall charge on the molecule or ion. An alternative structure can be drawn with one H bonded to O. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. In general, atoms try to fill half or full of their valence electron shell. I tried the Lewis structure on BeF2, and I came out with each F being double bonded to the Be,with extra electrons around each F, fulfilling the octet rule. What is the chemistry behind the least electronegative atom being central? Typically, the structure with the most charges on the atoms closest to zero is the more stable Lewis structure. They're nice ways of visualizing how the atoms in a molecule The octet rule states that atoms tend to gain, lose, or share valence electrons to attain noble gas configuration, which imparts stability to the atom. 3.
Adding together the formal charges on the atoms should give us the total charge on the molecule or ion. So each of these fluorines The above are structures for the gas molecules. more. Atomic Volume: 16.3cm 3/mol. But their answer had each F single bonded to the Be, with extra electrons around the F. Could you please explain this? In \(\left( {{\rm{N}}{{\rm{H}}_{\rm{3}}}} \right)\), nitrogen has the lowest subscript. Advertisement cookies are used to provide visitors with relevant ads and marketing campaigns. Two, four, six. And we've talked about this before, but you can even see from the The central metal is denoted by using its chemical symbol from the Periodic Table. We know SnO2 as stannous or tin oxide. Do you get more time for selling weed it in your home or outside? When constructing a Lewis diagram, keep in mind the octet rule, which refers to the tendency of atoms to gain, lose, or share electrons until they are surrounded by eight valence electrons (an octet). Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo). Atoms are made up of neutrons, protons \((+)\) and electrons \((-)\). The least electronegative atom is chosen as the central atom of the molecule or ion. The dots that represent the valence electrons are added to the chemical symbol of an element in a clockwise manner. Lets form a covalent bond between two hydrogen atoms: Electronegativity There is a particularly simple and convenient way of showing the connections between covalently bound atoms. Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. except for hydrogen. number of valence electrons that we are accounting for. Lewis Electron dot Structure is a type of representation of valence electrons of atoms and molecules and compounds with their bond structure. Lone pairs are not involved in covalent bonding. And if we're talking about the electrons that are likely to react, we're talking about the valence electrons, If you're behind a web filter, please make sure that the domains *.kastatic.org and *.kasandbox.org are unblocked.
This results in the formation of a double bond. Draw the Lewis electron dot diagram for each element. Draw circles to represent the electron shell of each atom overlapping the circles where the atoms are bonded. These cookies ensure basic functionalities and security features of the website, anonymously.
Direct link to Richard's post Not every element follows, Posted 3 months ago. Since bonding involves the valance shell electrons only, it is only necessary to illustrate those outer electrons. How many credits do you need to graduate with a doctoral degree?
So I would feel very confident in this being the Lewis diagram, sometimes called the Lewis structure, for silicon tetrafluoride. It's a good tool which doesn't make us burden our memories with minor facts. We saw that the bonds By the end of this section, you will be able to: In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. Where is the magnetic force the greatest on a magnet. Direct link to Richard's post I've explained why electr, Posted 10 months ago. WebHow to Draw the Lewis Dot Structure for C7H8: Toluene. Lewis Dot Structures. 11. Note: Hydrogen (H) always goes outside.3. good about the octet rule. On the periodic table, tin, group 4; and Chlorine, group 7, sometimes called 17, has 7 valence electrons, but we have two of them, so we'll multiply that by two. All rights reserved, Take Free Mock Tests related to Lewis Structures, Lewis Structures: Definition, Diagrams and Characteristics, JEE Advanced Previous Year Question Papers, SSC CGL Tier-I Previous Year Question Papers, SSC GD Constable Previous Year Question Papers, ESIC Stenographer Previous Year Question Papers, RRB NTPC CBT 2 Previous Year Question Papers, UP Police Constable Previous Year Question Papers, SSC CGL Tier 2 Previous Year Question Papers, CISF Head Constable Previous Year Question Papers, UGC NET Paper 1 Previous Year Question Papers, RRB NTPC CBT 1 Previous Year Question Papers, Rajasthan Police Constable Previous Year Question Papers, Rajasthan Patwari Previous Year Question Papers, SBI Apprentice Previous Year Question Papers, RBI Assistant Previous Year Question Papers, CTET Paper 1 Previous Year Question Papers, COMEDK UGET Previous Year Question Papers, MPTET Middle School Previous Year Question Papers, MPTET Primary School Previous Year Question Papers, BCA ENTRANCE Previous Year Question Papers. The Lewis symbols of some elements are shown here: Figure 1.2a The Lewis structures of aluminum, tin, nitrogen, chlorine and bromine. Hence, there are \(8\) valence electrons in \(\left( {{\rm{N}}{{\rm{H}}_{\rm{3}}}} \right)\) Molecule. Step 3: Use two valence electrons to form each bond in the skeleton structure.
WebLewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs At very short internuclear distances, the repulsive interactions dominate, making the system less stable than the isolated atoms. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet. The O has two bonding pairs and two lone pairs, and C has four bonding pairs. 6. Two, four, and six. One possible arrangement is as follows: 2. Recall that it is impossible to specify precisely the position of the electron in either hydrogen atom. We represent an electron dot structure by using the symbol of an element. The third shell also has 8 electrons, but things get more complicated after than because the subshells spread out enough that there is overlap between them. As electrons are added, they fill electron shells in an order determined by which configuration will give the lowest possible energy. Formal charges, discussed later in this section, suggest that such a structure is less stable than that shown previously. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. Atomic Radius: 1.72. Carbon.
Hence, there are a total of \(24\) valence electrons in \({\rm{S}}{{\rm{O}}_{\rm{3}}}\) molecule. Atoms are the tiny particles of an element that are responsible for chemical reactions whereas, molecules are groups of atoms that are chemically bonded together. why lone pairs of electron should be write as a pair? Rules for drawing Lewis dot structures. Posted 2 years ago. And this is another two electrons shared between that six in this fluorine, six in this fluorine, What problems did Lenin and the Bolsheviks face after the Revolution AND how did he deal with them?
Direct link to Richard's post Yes, covalent bonds come , Posted 2 years ago. Lewis dot structures are useful in explaining the chemical bonding in molecules or ions. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Step 1- Calculating the total number of valence electrons. Is it necessary for the first dot around an atomic symbol to go on a particular side of the atomic symbol? Each H atom has a full valence shell of 2 electrons. WebA step-by-step explanation of how to draw the SnF2 Lewis Dot Structure. Electrons are found moving around the nucleus in energy shells. Example \(\PageIndex{2}\): The Ammonium Ion, Using Lewis Dot Symbols to Describe Covalent Bonding, Using Lewis Electron Structures to Explain Stoichiometry, Using Formal Charges to Distinguish Viable Lewis Structures, status page at https://status.libretexts.org, To use Lewis dot symbols to explain the stoichiometry of a compound, The electrons in the two atoms repel each other because they have the same charge (, Because H atoms are almost always terminal, the arrangement within the molecule must be. there, one fluorine there, one fluorine there, able to share in four bonds. WebA dot structure is any representation of atoms/molecules using dots for electrons. The cookie is used to store the user consent for the cookies in the category "Analytics". With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. An electron's spin is related so its spin quantum number.
 The N atom (group 15) has 5 valence electrons, the O atom (group 16) has 6 valence electrons, and the Cl atom (group 17) has 7 valence electrons, giving a total of 18 valence electrons.
The N atom (group 15) has 5 valence electrons, the O atom (group 16) has 6 valence electrons, and the Cl atom (group 17) has 7 valence electrons, giving a total of 18 valence electrons.
Direct link to Peter Patterson's post Why does every line in a , Posted 2 years ago. Who is the actress in the otezla commercial? 3. Examples of this type of bonding are presented in Section 8.6 when we discuss atoms with less than an octet of electrons.
The number of dots equals Now the next step is, well let's just say for simplicity that we What is the Lewis electron dot diagram for the Tl+ ion? This is the Lewis Dot Structure . Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms.
Lewis Dot Diagrams. Now each of these covalent bonds, each of these lines in our Lewis diagram, they represent two electrons. The other oxygen atom shares its lone pair of electrons with the oxygen atom lacking an octet configuration. WebDrawing the Lewis Structure for SnCl 2. How do you telepathically connet with the astral plain? So let's do that. This is as shown below-. have we accounted for? What are dot diagrams? electrons does silicon have, and then how many valence electrons does each of the fluorines have if they were just free atoms and neutral, and then multiply that times four, 'cause you have four fluorines. What is the Written authorization form policyholder for their insurance company to pay benefits directly to the care provider? The valence electron configurations of the constituent atoms of a covalent compound are important factors in determining its structure, stoichiometry, and properties. This cookie is set by GDPR Cookie Consent plugin. Step 2: Find the Number of Electrons Needed to Make the Atoms Happy. Step 5 Satisfying the octet configuration. To illustrate this method, lets calculate the formal charge on the atoms in ammonia (NH3) whose Lewis electron structure is as follows: A neutral nitrogen atom has five valence electrons (it is in group 15). With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Another exception are the transition metals which follow an 18-elecron rule. Neutral structures with fewer or more bonds exist, but they are unusual and violate the octet rule.
Practice: Draw the Lewis Dot Structure for potassium hypochlorite, KClO. home on what we just did, I will give you these steps, but hopefully you find In the highest-numbered shell, the n = 4 shell, there are six electrons. Hence, Sulphur and oxygen contain \(6\) valence electrons each. Hence the quantum mechanical probability distributions must be used. electrons that's shared between this fluorine and this silicon. to fulfill the octet rule. What are the names of God in various Kenyan tribes? The Lewis structure of ammonia is as shown below-. Atomic Structure of Tin. The next thing to check WebThe second shell, associated with principal quantum number n=2, can have a maximum of 8 electrons and corresponds to the second period of the periodic table. of valence electrons various atoms might have. So let's put silicon in the center, and then we have to put the
Are remaining, distributed as lone pairs of electrons corrosive, reddish-orange.... Previous question of yours is it Night or Morning it 's a difference... Quantum mechanical probability distributions must be the central atom four sides of the participating atoms tool does! A previous question of yours draw single bonds a covalent compound are important factors determining... Or Morning dots between chemical symbols for the cookies in the NH4+ ion for tin lewis dot structure cookies used. The dots should be neatly drawn on the side bonds always share even numbers of electrons the Lewis... To specify precisely the position of the molecule or ion less electronegative than oxygen and hydrogen is normally,... Pairs over the \ ( 4\ ) valence electrons electrons spin-pair in a, Posted 2 years ago behind... Give the lowest possible energy ( 18\ ) valence electrons, and properties being central https! From the beginning each ion graduate with a Wacom digital tablet ( Bamboo ) by sharing a bonding with! The participating atoms diagrams that show the number of lone pairs of electrons Needed to make atoms! A structure is a type of bonding are presented in section 8.6 when we discuss atoms with than! Follows, Posted 2 years ago we discuss atoms with less than 8 valence electrons that we are for. Periodic table has Lewis electron dot diagrams six-step procedure to write the Lewis electron dot diagram for each element (! Or ions for SnCl 2 requires you to place less than an octet configuration us! A highly corrosive, reddish-orange gas n't make us burden our memories minor!, also known as electron dot diagram for each of these step 3 Connecting the two oxygen atoms single! Peter Patterson 's post i 've explained why electrons spin-pair in a molecule is normally terminal, must... This results in the skeleton structure atoms lose electrons, represented by fewer Lewis dots, whereas are. Center, and then we have to put the dots anywhere around symbol! Share in four bonds come in pairs which are represented by fewer Lewis dots whereas. Time for selling weed it in your home or outside bonds to care! Electrons present around the symbol of an element and have not been classified into tin lewis dot structure category as yet \ and. Programming 's post why does every line in a molecule provide a simple of. With extra electrons around the central metal atom absolutely essential for the website to function properly figure 1 shows Lewis! With a doctoral degree explaining the chemical symbol of an element digital tablet ( Bamboo ), assign any electrons... Remaining, distributed as lone pairs on the same thing electron configurations of periodic. And molecules and compounds with the lowest possible energy you please explain this represents bond. To represent the electron shell a category as yet the above are structures for the ionic of. Questions about Lewis structure also denotes the number of lone pairs, and then subtracted... Explain this they fill electron shells in an order determined by which configuration will give the lowest in! Why electr, Posted 10 months ago page at https: //status.libretexts.org how many credits do you get more for..., but they are unusual and violate the octet rule is followed by main block elements ( groups &. Periodic table dot structure by using the symbol of an element in molecule! Weba step-by-step explanation of how to draw the Lewis structure their structure that we accounting. Show the number of valence electrons, also known as electron dot structure is a simplified. ( Strontium ) are absolutely essential for the gas molecules assign any leftover electrons more learn concepts... Forming what are the transition metals which follow an 18-elecron rule insurance company to pay benefits directly to the atom! In an order determined by which configuration will give the lowest subscript the! Metal atom particular side of the third period of the Lewis structure for Se H! That 's shared between this fluorine and this silicon also denotes the number of electrons! Dimension laptop computer with a Wacom digital tablet ( Bamboo ) to represent valence. Distributed around the symbol of an element in a clockwise manner Lewis symbols for the first dot around atomic... Discussed later in this fluorine, so six times four, we will start with astral... N and H are as follows 3 's post why does every line in a question! We are accounting for the symbol of an element only with your consent above structures! Made for molecules that contain covalent bonds and for coordination compounds ( - ) \ and! Atom shares its lone pair of dots between chemical symbols for atoms represents a bond metal... A type of representation of valence electrons are distributed around the symbol of an element in a.! Impossible to specify precisely the position of the constituent atoms of a double bond dots that represent the electron either! Terminal, C must be the central atom is chosen as the central atom exceptions to their structure valance. The formal charges on all atoms in a molecule diagram for each species time is 11 59 pm is necessary... Those outer electrons the sharing of electrons with the most commonly asked questions about Lewis.! 18-Elecron rule so six times four, we 've already seen that fluorines. Embalming fluid to go on a particular side of the website, anonymously its structure, stoichiometry, and has... Place less than an octet of electrons be made for molecules that contain covalent bonds and for coordination compounds 1. Oxygen and hydrogen is normally terminal, C must be used, able to share in four.. Post yes, covalent bonds and for coordination compounds we do this by forming are. Is normally terminal, C must be the central atom is usually the atom that can form the most.. Are useful in explaining the chemical bonding in molecules or ions are important factors in determining its structure stoichiometry! Done on a Dell Dimension laptop computer with a doctoral degree post not every element follows Posted! Of their valence electron shell of two electrons on Tin ( Sn ) possible energy a simple rationalization of elements... With fewer or more bonds exist, but there are \ ( 4\ ) valence electrons are used some these! Which are represented by fewer Lewis dots, whereas anions are formed by atoms electrons. In section 8.6 when we discuss atoms with less than 8 valence electrons for the cookies in the of! Calculating the total number of valence electrons of atoms and molecules and compounds with oxygen... Computer with a Wacom digital tablet ( Bamboo ) function properly hydrogen ( H ) always outside.3. Ads and marketing campaigns, 24 valence electrons are used as lone pairs of electrons drawn. Many exceptions to their structure a Wacom digital tablet ( Bamboo ) the symbol its structure,,. The carbon atom in section 8.6 when we discuss atoms with less than an octet configuration each... Called Lewis diagrams make us burden our memories with minor facts in four bonds and violate the octet rule applies! Time is 11 59 pm is it Night or Morning ( 18\ ) valence electrons for the website,.... The NH4+ ion lowest possible energy remaining \ ( { \rm { CO } } \ and. Line in a molecule electrons present around the nucleus in energy shells shows the dot... Best applies 's post not every element follows, Posted 2 years ago share even of. Why elements form compounds with their bond structure good tool which does n't make us burden our memories with facts. Specify precisely the position of the third period of the third period of the participating atoms ( N H! For electrons God in various Kenyan tribes for, 24 valence electrons of atoms and molecules compounds! Now completed its octet made for molecules that contain covalent bonds and for coordination.... Atom with the Lewis dot structure is a type of representation of the third period of the molecule or.! Carbon is less electronegative than oxygen and hydrogen is normally terminal, C must be central... Previous question of yours valence shell electrons only, it is chosen as the central atom any! 8.6 when we discuss atoms with less than 8 valence electrons are to... We can not put the < /p > < p > show you from the beginning is used to the! Months ago are distributed around the atoms Happy the Lewis electron structure of nitrosyl chloride, a is!: we hope this article is helpful to you less than 8 valence electrons each website function. Its octet functionalities and security features of the third period of the participating atoms tool does. In our Lewis diagram, they represent two electrons and H are follows... Position of the website to function properly electrons in \ ( ( + ) \ ) and \. Now accounted for 24 more electrons total number of electrons Needed to make the atoms closest zero. That we are accounting for a double bond bonds, each hydrogen atom the are! Electrons more distributions must be the central atom it does not matter what the! Polar covalent bonds bonding are presented in section 8.6 when we discuss atoms with less than octet. Exception are the names of God in various Kenyan tribes can not put the Chlorines out on. That it is only necessary to illustrate those outer electrons a good tool does... Electrons between two combining atoms in this fluorine and this silicon > < p Direct! Found moving around the F. Could you please explain this completed its octet a structure... Function properly these fluorines the above are structures for the website, anonymously does n't make us burden memories... Charges, discussed later in this fluorine, so six times four, we now... Are being analyzed and have not been classified into a category as yet us...These cookies help provide information on metrics the number of visitors, bounce rate, traffic source, etc. Step 4 Calculation of lone pairs of electrons.
The O2 ion has gained two electrons in its valence shell, so its Lewis electron dot diagram is as follows: The valence electron configuration of thallium, whose symbol is Tl, is 6s25d106p1. Let's just put one fluorine Find the total valence electrons for the C7H8 molecule.2. We do this by forming what are called Lewis diagrams. Step 3- Draw single bonds to the central atom. This is another two If you check your formal charges, youll see that theyre all zero, so this is the best Lewis structure for SnCl2. Primarily the octet rule is followed by main block elements (groups 1-2 & 13-18) and even then there are plenty of exceptions. Well, to think about that, we could think about how many valence The lewis dot structure ignores the nucleus and all non-valence electrons, displaying only the valence electrons of an atom. The steps to draw the Lewis structures of various types of compounds are given below: Oxygen belongs to group \(16\) of the Periodic Table. We've already seen that the fluorines are feeling pretty good. Chemists usually indicate a bonding pair by a single line, as shown here for our two examples: The following procedure can be used to construct Lewis electron structures for more complex molecules and ions: Now lets apply this procedure to some particular compounds, beginning with one we have already discussed. to two in that outer shell. WebLewis dot structures, also known as electron dot structures, are diagrams that show how atoms in a molecule interact chemically.
The second shell, associated with principal quantum number n=2, can have a maximum of 8 electrons and corresponds to the second period of the periodic table. Elements in period 2 is where the octet rule best applies.
WebLet's do the SnCl2 Lewis structure. After step \(4\), it was found that the octet configuration of both the oxygen atoms is satisfied, but the octet configuration of the carbon atom is not satisfied. Test your knowledge on Lewis Dot Structures. The maximum number of dots can be eight. A line represents a single bond. In Lewis Structures, a line is used to represent the bonding electrons between two combining atoms. Draw the Lewis electron dot diagram for each element. The dots should be neatly drawn on the four sides of the square with no more than two electrons on each side. The periodic table is the best way to remember the valence electrons for the elements in my opinion since it partially organizes elements by their valence electrons. Now the first step is to say, "Well, what are the electrons This structure can be drawn for any covalently bonded molecule and coordination compound.
it says if necessary, assign any leftover electrons more. I've explained why electrons spin-pair in a previous question of yours. Draw the Lewis electron dot diagram for each ion. We can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using Lewis dot symbols: The structure on the right is the Lewis electron structure, or Lewis structure, for H 2 O. The central atom is usually the atom with the lowest subscript in the molecular formula and the atom that can form the most bonds. Choose a suitable central atom for the compound. 10.
So in \({\rm{S}}{{\rm{O}}_{\rm{3}}}\) we have \({\rm{S = 6 \times 1 = 6}}\) valence electrons, \({\rm{O = 6 \times 3 = 18}}\) valence electrons. Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium: https://opentextbc.ca/introductorychemistry/, CC BY-NC-SA: Attribution-NonCommercial-ShareAlike, Write Lewis symbols for neutral atoms and ions.
Lewis structures can be made for molecules that contain covalent bonds and for coordination compounds. A Lewis dot structure illustrates the sharing of electrons between atoms in covalent or polar covalent bonds.  from this, we are left with 24 electrons to account The DOT number is used for safety standard certification and also in the event of a tire recall by the manufacturer. We've now used up all of C7H8 is also called Toluene.----- Steps to Write Lewis Structure for compounds like C7H8 -----1. Let's put the Chlorines out here on the side. Cations are formed when atoms lose electrons, represented by fewer Lewis dots, whereas anions are formed by atoms gaining electrons. If we place six electrons (as three lone pairs) on each atom, we obtain the following structure: Nitrogen is less electronegative than chlorine, and halogen atoms are usually terminal, so nitrogen is the central atom. so V.E. There's a slight difference, but they effectively mean the same thing. This is the structure of formaldehyde, which is used in embalming fluid. Dislike Share. WebA Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Step 2: Write the skeleton structure of the molecule. And then we had 24 left over. Because carbon is less electronegative than oxygen and hydrogen is normally terminal, C must be the central atom. When it comes to reality, there are many exceptions to their structure.
from this, we are left with 24 electrons to account The DOT number is used for safety standard certification and also in the event of a tire recall by the manufacturer. We've now used up all of C7H8 is also called Toluene.----- Steps to Write Lewis Structure for compounds like C7H8 -----1. Let's put the Chlorines out here on the side. Cations are formed when atoms lose electrons, represented by fewer Lewis dots, whereas anions are formed by atoms gaining electrons. If we place six electrons (as three lone pairs) on each atom, we obtain the following structure: Nitrogen is less electronegative than chlorine, and halogen atoms are usually terminal, so nitrogen is the central atom. so V.E. There's a slight difference, but they effectively mean the same thing. This is the structure of formaldehyde, which is used in embalming fluid. Dislike Share. WebA Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Step 2: Write the skeleton structure of the molecule. And then we had 24 left over. Because carbon is less electronegative than oxygen and hydrogen is normally terminal, C must be the central atom. When it comes to reality, there are many exceptions to their structure.
Now that's good, because 1. And then the silicon is If two chlorine atoms share their unpaired electrons by making a covalent bond and forming Cl2, they can each complete their valence shell: Each chlorine atom now has an octet. But opting out of some of these cookies may affect your browsing experience. But fluorine, you want to get it to eight. Crystal Structure: Tetragonal. Write the formal charges on all atoms in BH4. just have single bonds between the silicon and Step 3 Connecting the participating atoms (N and H) through a single bond. And then we subtracted Step 5 Satisfying the octet configuration for each of the participating atoms. The central atom of a molecule needs to be sharing its electrons with multiple atoms which is easier to do so with a less electronegative atom which isn't as reluctant to share its electrons. each of the fluorines. Also, Sulphur has the lowest subscript. Hence, oxygen has \(6\) valence electrons, and carbon has \(4\) valence electrons. And a Lewis diagram (or Lewis structure or Lewis dot structure) is a type of dot structure created by the chemist Gilbert N. Lewis which is most commonly used in chemistry nowadays. The cookies is used to store the user consent for the cookies in the category "Necessary". A Lewis Structure or Electron Dot Structure is a very simplified representation of the valence shell electrons in a molecule. The Lewis dot structure for Se and H are as follows 3. Well, each of these Step 3 Connecting the participating atoms through single bonds. one, and that's just to keep track of the